Description
20 KN95 Masks (bundle comes with two packages of 10 KN95 face masks):
KN95 face masks are made by an FDA-registered manufacturer. Please note that GB2626-2006 on the front of the package is the code of regulation in China for KN95 masks.
See a comparison chart from 3M on the difference between N95 masks and KN95 masks.
See filtration test results for the masks here. Our KN95 masks have a waterproof layer in them (seen in photos of mask holding water but not getting wet on the opposite side of the mask) that protects both the wearer and those on the other side of the mask.
While these masks have passed filtration tests, these masks are not medical grade and not intended for use in hospitals. This is why the packaging says “This product is a non-medical device.”
True Spin Dental is neither affiliated with, nor endorsed by, the manufacturer listed on this page. See Disclaimer Page for details.
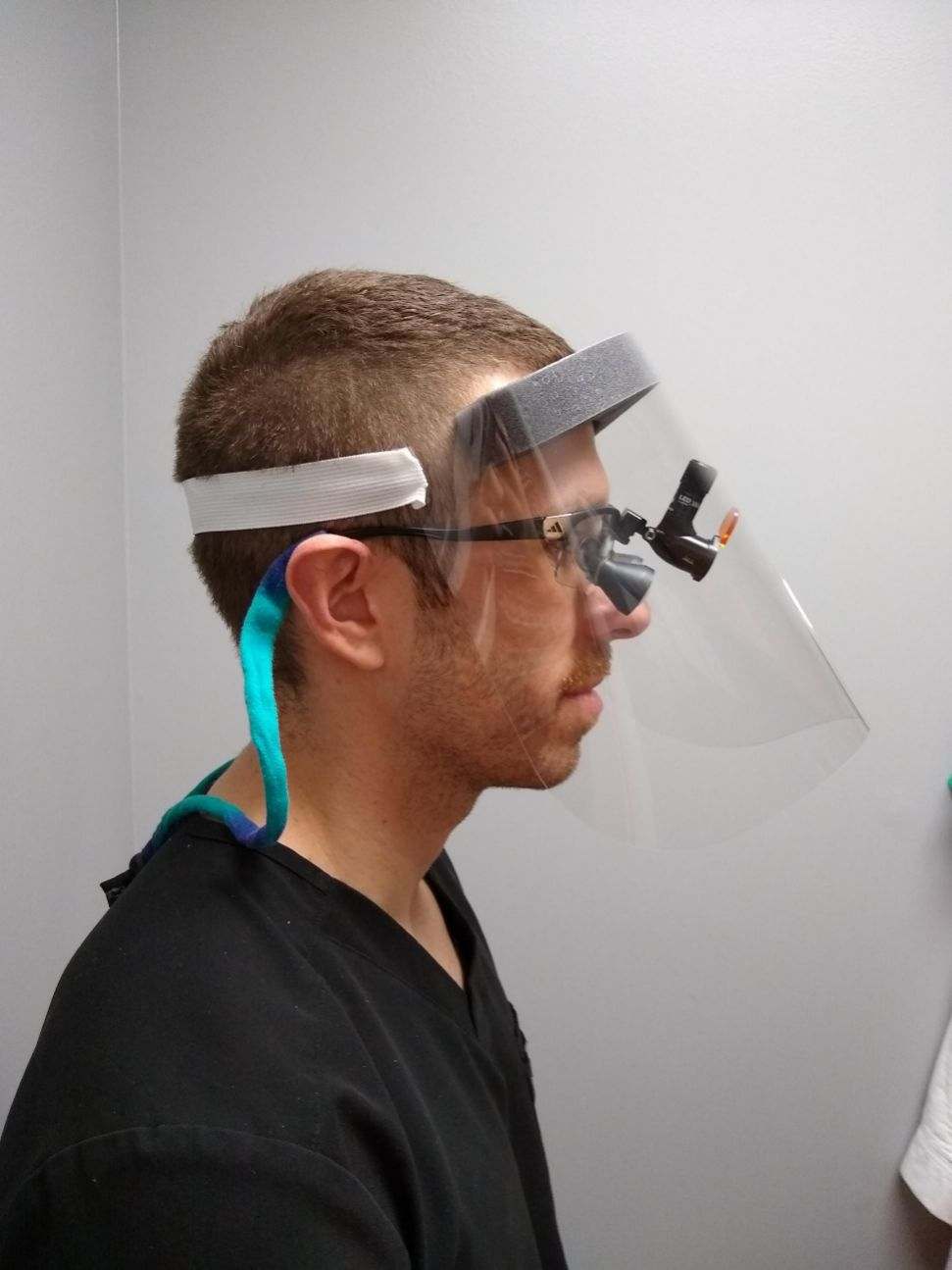
10 Face Shields
The ADA is recommending face shields and masks be used when treating patients, in addition to a mask, “to protect mucous membranes of the eyes, nose, and mouth during procedures likely to generate splashing or spattering [(large droplets)] of blood or other body fluids.” (ADA, quoted from https://www.cdc.gov/mmwr/PDF/rr/rr5217.pdf)
Materials
- Clear Shield Material: Clear Polyethylene (PET) Film (0.012″ thick)
- Elastic Headband Material: Elastic Band (1″W x 13″ Long), Latex-Free, White / Grey / Black
- Brow Foam Material: Adhesive Backed Polyurethane Foam (1.375″ thick)
Face shields are NOT manufactured in a sterile environment. Face shields are NOT FDA approved, but the FDA will not object to their use during the COVID-19 pandemic. See FDA guidance: https://www.fda.gov/media/136449/download
This face shield was designed, sourced, manufactured, and assembled in the USA.