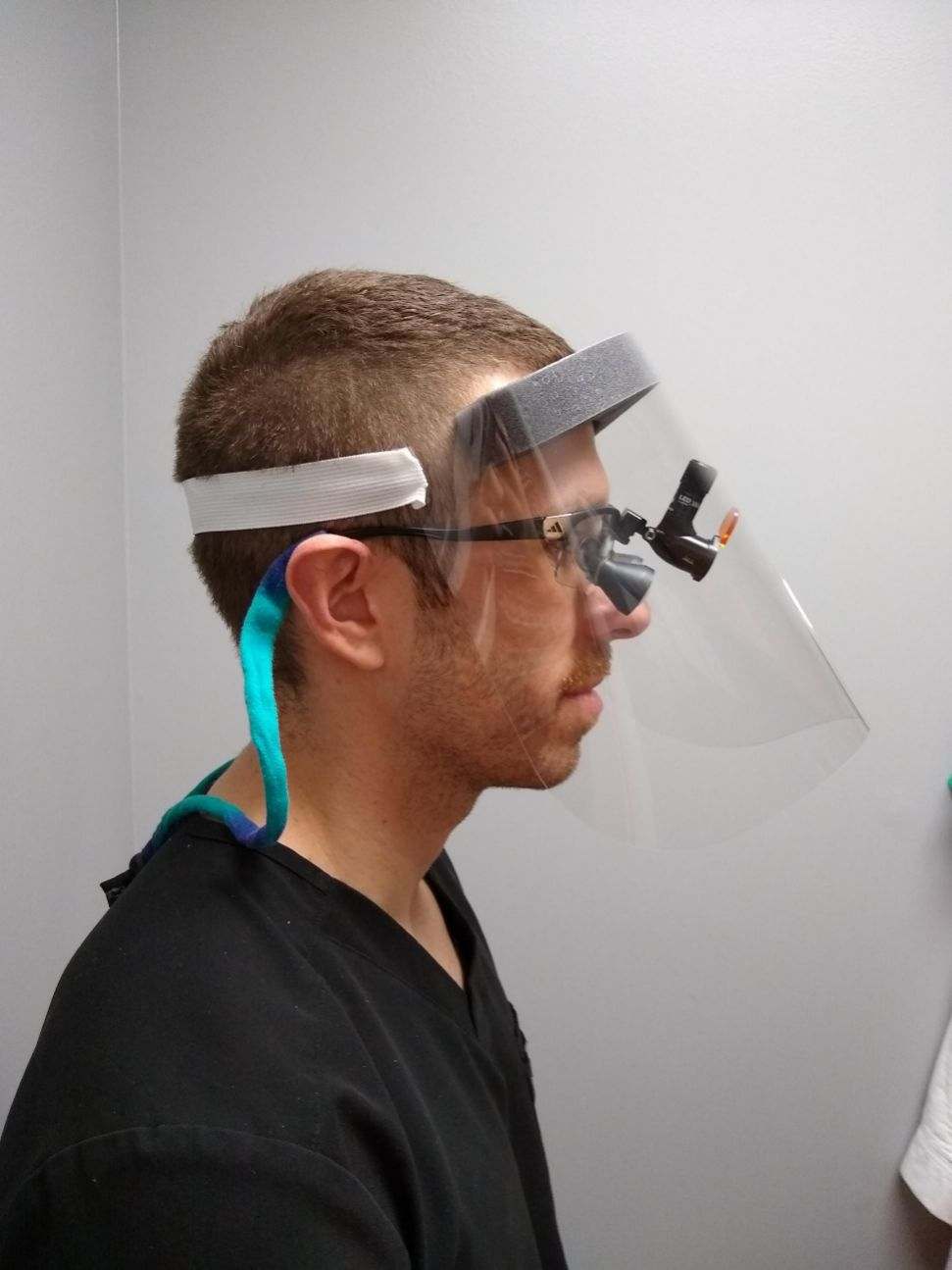
Description
The ADA is recommending face shields and masks be used when treating patients, in addition to a mask, “to protect mucous membranes of the eyes, nose, and mouth during procedures likely to generate splashing or spattering [(large droplets)] of blood or other body fluids.” (ADA, quoted from https://www.cdc.gov/mmwr/PDF/rr/rr5217.pdf)
Materials:
- Clear Shield Material: Clear Polyethylene (PET) Film (0.012″ thick)
- Elastic Headband Material: Elastic Band (1″W x 13″ Long), Latex-Free, White / Grey / Black
- Brow Foam Material: Adhesive Backed Polyurethane Foam (1.375″ thick)
Face shields are NOT manufactured in a sterile environment. Face shields are NOT FDA approved, but the FDA will not object to their use during the COVID-19 pandemic. See FDA guidance: https://www.fda.gov/media/136449/download
This face shield was designed, sourced, manufactured, and assembled in the USA.
True Spin Dental is neither affiliated with, nor endorsed by, the manufacturer listed on this page. See Disclaimer Page for details.